Mendeleev made many other contributions to chemistry, but was most widely known for the periodic table. In modern times, we can use the periodic table to learn a lot of information about each element, as well as to make predictions about how elements will behave in certain situations. Scientists also discovered the links between atomic mass, the underlying structure of atoms, and the way elements behave. 
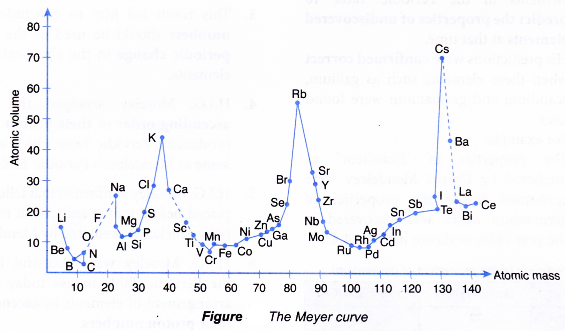
As scientists continued to learn more about the atom, they learned that not just atomic mass and number, but other properties, like atomic size and atomic structure, also changed gradually from left to right on the periodic table. Mendeleev knew that the atomic mass increased from left to right and assigned numbers to the elements based on his periodic table. There are trends in the elements from left to right on the periodic table. The name “periodic table” refers to the periodic nature of the arrangement of the elements. The periodic table is more than just a table of known elements. Illustration by Dmitri Mendeleev, courtesy of Den fjättrade ankan from Wikipedia At the time, the underlying chemical structure of atoms was not known. In his first periodic table Mendeleev arranged the elements by their mass and other chemical properties. Mendeleev’s fist attempt to organize the elements is seen in SF Fig. While working on his textbook from 1868–1870, he began formulating ideas for a way to organize the elements. Petersburg, Mendeleev felt that there were no adequate textbooks for teaching chemistry, so he began to write his own. While teaching chemistry as a professor at the University of St. At university, he earned an advanced degree in chemistry while training as a teacher. As a child he learned about chemistry in his mother’s glassblowing factory. Mendeleev was interested in many fields of science and studied a wide variety of science topics throughout his life. He was the youngest of 13 (or 11, 14, or 17, depending on the source) children. Mendeleev has been called the “father of the periodic table”.ĭmitri Mendeleev was born in 1834 in Russia. Meyer’s periodic table was very similar to Mendeleev’s, but because it was published after Mendeleev’s, Mendeleev got most of the credit. In the late 1800s, Lothar Meyer was developing a periodic table at the same time as Mendeleev. Newlands’ table was not well received because two elements were in the same box in several spots on his table. John Newlands’ table of elements followed the Law of Octaves (1863). 2.11), proposing the elements followed the Law of Octaves. In 1863, John Newlands published a table with elements arranged in groups of eight (SF Fig. 
On this three-dimensional table, the elements lined up in vertical columns. Beguyer de Chancourtois arranged the elements on a cylinder, with the elements wrapping around the cylinder. Dobereiner discovered more of these sets of three and called it the Law of Triads. These three elements had similar properties. In 1817, Johann Dobereiner noticed that the atomic mass of strontium was halfway between those of calcium and barium.From 1817 through the time of Mendeleev, many scientists made important contributions to what would become the modern periodic table. As more information was learned about individual elements, scientists wanted to organize the elements in a useful way. They learned many other chemical properties by methodically exposing individual elements to various chemicals and observing the reactions. During this time, scientists determined the masses of the individual elements. Many more elements were discovered over the next 220 years. Many, like gold, silver, tin, copper, lead, and mercury had been known since antiquity. When scientists began to classify and organize the elements, about 63 elements were known.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
